Environment & Energy
Related: About this forumEthanol and unspecified alkenes via the hydrogenation of CO2 with earth abundant catalysts.
I came across a paper this evening which while somewhat sketchy - in the sense that one would like more details - caught my eye inasmuch as I'm always attracted to systems that use readily available elements as opposed to those that rely on exotic and/or expensive elements. I won't spend much time with the paper, but I just thought it worth mentioning it, since the catalyst is a mixed catalyst with three very common elements, copper, iron, and sodium.
It's this paper:
Synthesis of Alkene and Ethanol in CO2 Hydrogenation on a Highly Active Sputtering CuNaFe Catalyst Zhiyan Si, Linkai Wang, Yu Han, Jiafeng Yu, Qingjie Ge, Chunyang Zeng, and Jian Sun ACS Sustainable Chemistry & Engineering 2022 10 (45), 14972-14979.
The language of the paper shows some artifacts of having been translated from the Chinese by a non-native English speaker, but it's intriguing in any case.
From the introduction to the paper:
For the above chemicals, olefin and ethanol are both high-value intermediates or chemicals for modern society. The synthesis of olefin via CO2 hydrogenation generally requires a tandem reaction of C–O activation, as well as subsequent C–C coupling. (38−41) The representative catalyst is composed of an oxides/zeolite composite with methanol as an intermediate, and an iron-based catalyst with CO as an intermediate. In comparison, producing ethanol is more challenging to achieve from CO2 hydrogenation due to the complexity in various reaction pathways and the uncontrollability on C–O insertion in parallel with C–C coupling from untamed surface sites. To date, a series of noble metal catalysts, modified Co-based catalysts, Cu-based catalysts, and Mo-based catalysts with or without promoters have been successfully developed in fixed-bed or tank reactors. (12,42−44) However, the common problem for both synthesizing olefin and ethanol lies in the lower efficiency in a single pass process, especially in terms of a low carbon utilization rate under a high space velocity on catalysts, which severely limits the value in potential industrial application. Considering the state-of-the-art process, the coproduction of olefin and ethanol is meaningful and favorable to the chemical industry in view of the facile separation between gas (mainly C2–C4 lower olefin) and liquid (ethanol) product. But unfortunately, simultaneous synthesis of the above two high-value products from CO2 and H2 in one efficient step is very challenging owing to the requirement of precisely designing active sites. It generally requires a synergistic process including C–O activation, C–C coupling, and C–O insertion on a well-defined catalyst.
The physical sputtering apparatus designed by Abe, Tsubaki, et al. and our group was recently employed for synthesizing heterogeneous catalysts by means of the unique properties of sputtered metal nanoparticles. The various sputtered metals on supports show obvious merits, such as high activity, feasibility without high-temperature reduction, and noble-metal-like properties in the classic F-T synthesis, ester hydrogenation, methanation, and reverse water gas shift reaction. (45−48) It provides potential for the precise design of active sites in heterogeneous catalysis.
Herein, we report a well-defined ternary catalyst comprising highly active Cu nanoparticles sputtered on a Na modified Fe3O4 support (sp-CuNaFe), which is prepared by a self-made physical sputtering apparatus (Supporting Information, Figure S1). In CO2 hydrogenation, it shows a remarkable promotion in simultaneously synthesizing olefins and ethanol compared to other conventional Fe and CuFe series catalysts. The total space time yield (STY) rate of high-value olefin and ethanol can reach as high as 833 mg·g–1·h–1 at a mild condition of 310 °C and 3 MPa, which ranked as one of the best performances among related studies. The key to success is the creation of the coordination of well-dispersed sputtered Cu nanoparticles with surrounding NaFe, providing a suitable distance for C–C coupling and C–O insertion...
Unfortunately the generic popular term "olefin" (the proper term is "alkene" ) doesn't specify which alkenes are produced; it is one thing if it's ethene (ethylene), another if it a mixture of higher alkenes. Nevertheless all alkenes can be polymerized, and in this sense they represent fixed carbon.
The authors do not seem to use - perhaps because it's not available in their labs - GC/MS or GC/MS/MS; they rely on GC-FID, which perhaps accounts for their failure to identify the components of the mixture better.
Here's a figure from the text:
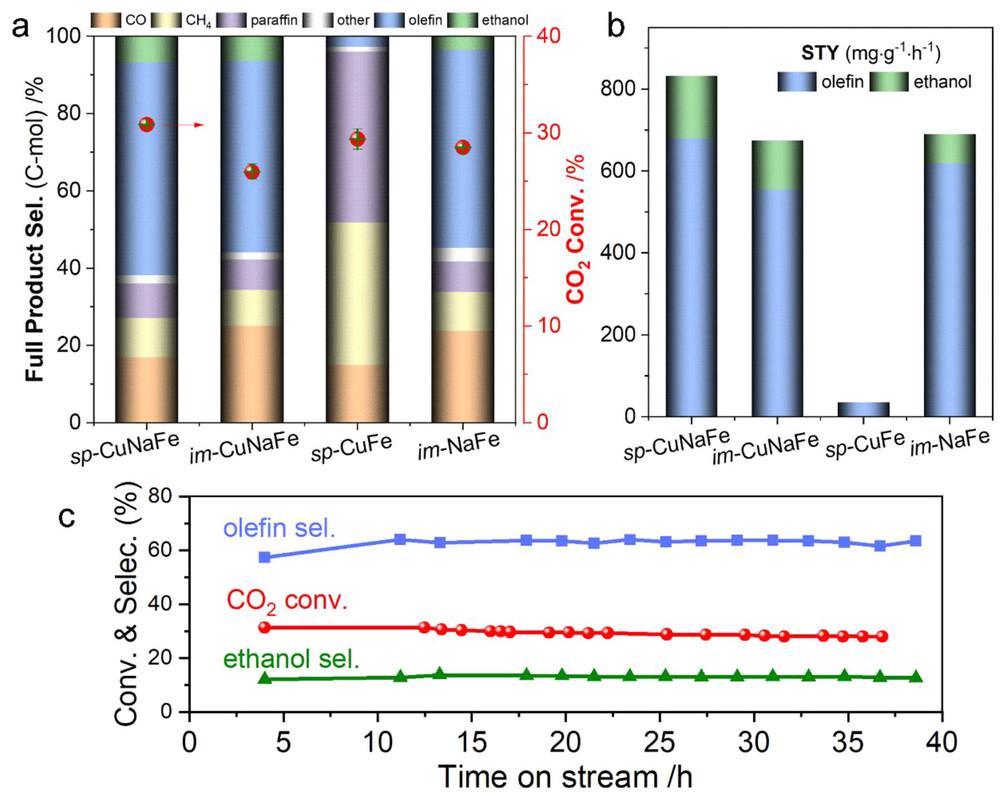
The caption:
The yield is low, and the product distribution is, again, unclear. But this is potentially a very cheap and very sustainable catalyst.
Still, one hopes the paper gets some attention and the work is subject to efforts at optimization.
While I have no use for the idea of hydrogen as a consumer fuel - a dumb idea that happily goes nowhere - it is a very useful captive intermediate and may be produced either by the thermochemical splitting of water using thermochemical cycles, or by carbon dioxide splitting followed by the water gas reaction, in which case carbon dioxide is the catalyst used for splitting water. Such a procedure offers the possibility of high efficiency and the recovery of exergy from high temperature systems, an effort that should be the focus of engineering at a breakneck speed. We are out of time.
Enjoy the workweek as it winds down.
eppur_se_muova
(36,256 posts)Most of the mass is in low carbon numbers, sort of like some petroleum cracking. This stream could likely be fed straight into a standing petrochemical plant.
There's a handy summary table showing several CO2 hydrogenation catalysts from the literature. Almost a review article in itself.
NNadir
(33,509 posts)...the catalyst involved readily available elements with relatively low environmental impact.
(I seldom even scan papers into papers whose titles involve cobalt catalysts, for example.)
NNadir
(33,509 posts)...I can certainly see places, given the likely low cost of the catalyst, where it could be useful.
I'm actually an MeOH/DME kind of guy in any case and for me the holy grail, partially realized, is direct hydrogenation of carbon dioxide to DME.
The alkenes produced according to the SI information may have nice uses particularly in synthesis.
I would like to see people play around with this type of catalyst.